 Although infection with one DENV serotype confers lifelong homotypic immunity, heterotypic cross-protection is timely limited. Thereby, direct methods (virus culture, real-time PCR, antigen detection) as well as (with limitations) IgM detection can be used for diagnosis of acute infections while the presence of DENV-specific IgG antibodies indicates a prior exposure to DENV. Laboratory diagnostics of DENV infection can be based on the detection of the virus itself (by virus culture), viral RNA (by real-time PCR), viral antigen (by ELISA or lateral flow testing) and the detection of a virus specific IgM/IgG response. In an attempt to adapt the system better to the clinical situation this classification has been revised in 2009 and is since then replaced by the categories “dengue infection with warning signs”, “dengue infection without warning signs” and “severe dengue infection”. Historically, cases have been classified according to WHO guidelines in the categories “dengue fever” and “dengue hemorrhagic fever”, the latter including also the most severe grade, the dengue shock syndrome. Nevertheless, some patients develop severe symptoms caused by increased vascular permeability, plasma leakage and intravascular volume depletion that can be lethal if left untreated. Upon defeverescence, usually occurring between days 3–7 of illness, most patients start to recover from the disease. During this febrile phase, leucopenia and thrombocytopenia are frequently observed this finding (as the occurrence of petechiae and/or a positive tourniquet test) can support differentiation of DF from other febrile illnesses. In an estimated 25% of infected individuals, a sudden onset of high-grade fever occurs after an incubation period of 4–7 days, often accompanied by other unspecific disease symptoms like headache, myalgia, arthralgia, nausea, vomiting, and rash. As in other flavivirus infections, asymptomatic or sub-clinical infection is frequent. Transmission of the four genetically distinct DENV serotypes mainly occurs by the bite of infected Aedes aegypti mosquitoes, limited vectorial capacity of Aedes albopictus has also been shown. Globalization and climate change facilitate the geographic spread of both the mosquito vector and the pathogen to previously unaffected areas. In endemic countries, disease burden has increased dramatically over the recent years due to population growth and urbanization. 
With an estimated incidence of 100 million clinically apparent cases per year in Asia, Latin America and Africa, dengue fever is the most frequent arboviral disease globally. While a good concordance (Cohen’s kappa 0.78) was found between the SD Bioline Dengue Duo NS1 test and the Platelia NS1 antigen ELISA, both the SD Bioline Dengue Duo IgM and IgG test displayed a significantly lower sensitivity than the corresponding ELISA tests. Diagnostic sensitivity of the SD Bioline Dengue Duo NS1 test strongly correlated with viral load, decreased rapidly over the acute phase of the disease, and was significantly reduced in presence of high anti-DENV IgG antibody titers resulting from secondary DENV infection. 57.6%, 22.8% and 44.6% of acute phase serum samples tested positive in the SD Bioline Dengue Duo NS1, IgM, and IgG test, respectively.  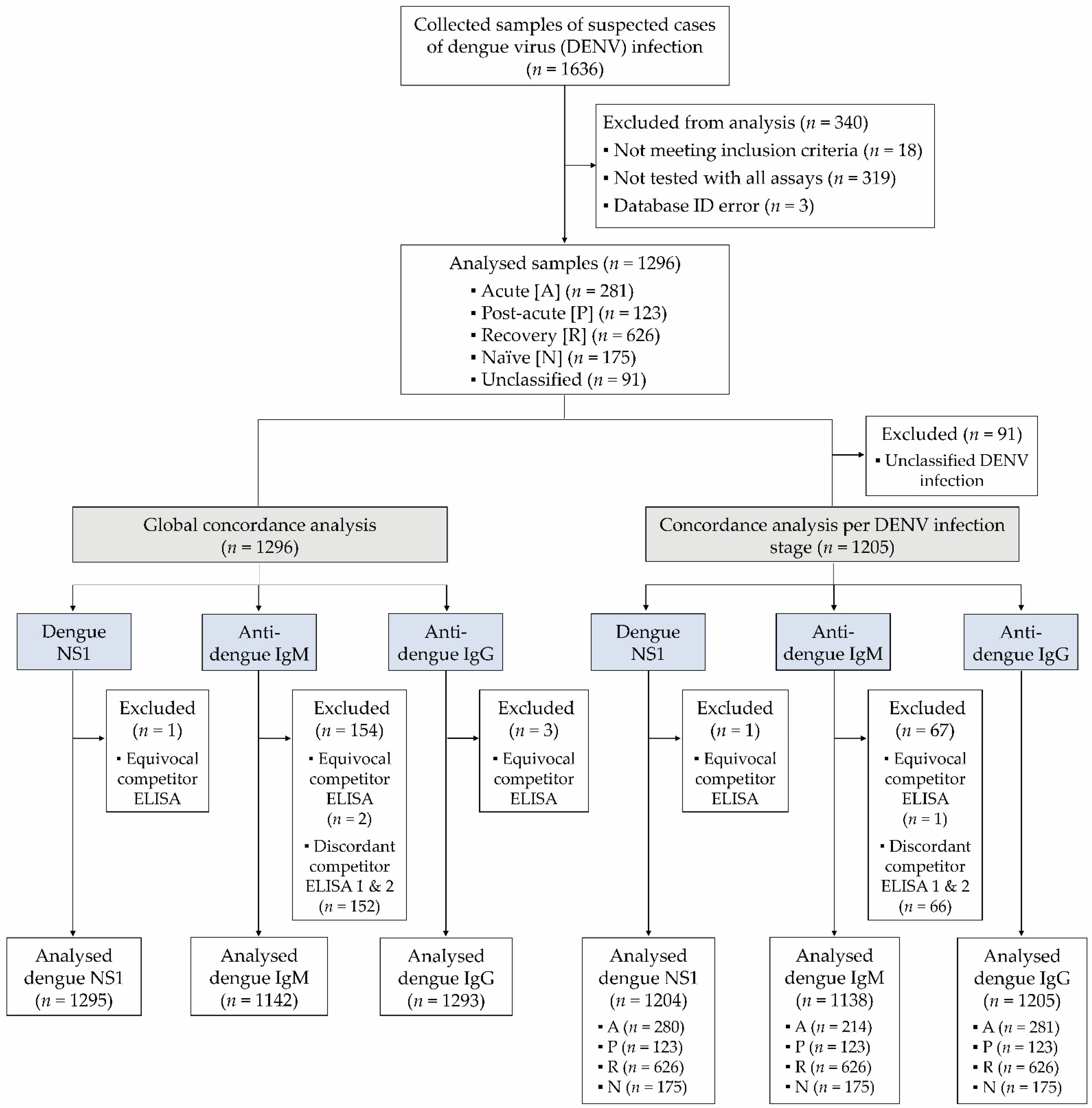
IgM seroconversion was assayed using follow-up samples of 35 patients collected in the convalescent phase. A subset of 74 samples was additionally tested with the Platelia NS1 antigen test, the Panbio DENV μ-capture ELISA and the Panbio DENV IgG ELISA. Jörg Blessmann, Conceptualization, Data curation, Formal analysis, Investigation, Supervision, Writing – review & editing, 1 Yvonne Winkelmann, Investigation, Writing – review & editing, 2 Latdamone Keoviengkhone, Investigation, Writing – review & editing, 3 Vatsana Sopraseuth, Investigation, Writing – review & editing, 3 Simone Kann, Investigation, Supervision, Writing – review & editing, 4 Jessica Hansen, Investigation, Writing – review & editing, 2 Hussein El Halas, Conceptualization, Investigation, Methodology, Writing – review & editing, 5 Petra Emmerich, Investigation, Supervision, Writing – review & editing, 6, 7 Jonas Schmidt-Chanasit, Investigation, Supervision, Writing – review & editing, 8, 9 Herbert Schmitz, Investigation, Supervision, Writing – review & editing, 6 Angela Mika, Formal analysis, Investigation, Supervision, Writing – review & editing, 2 and Christina Deschermeier, Conceptualization, Data curation, Formal analysis, Investigation, Supervision, Visualization, Writing – original draft 2, *ĩ2 acute phase serum samples from patients with a PCR-confirmed DENV infection collected in Lao People’s Democratic Republic (Lao PDR) in 20 were analyzed with the SD Bioline Dengue Duo test. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
